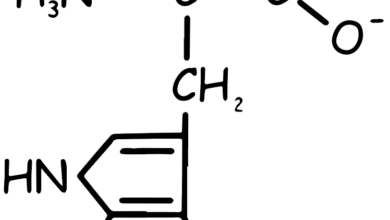
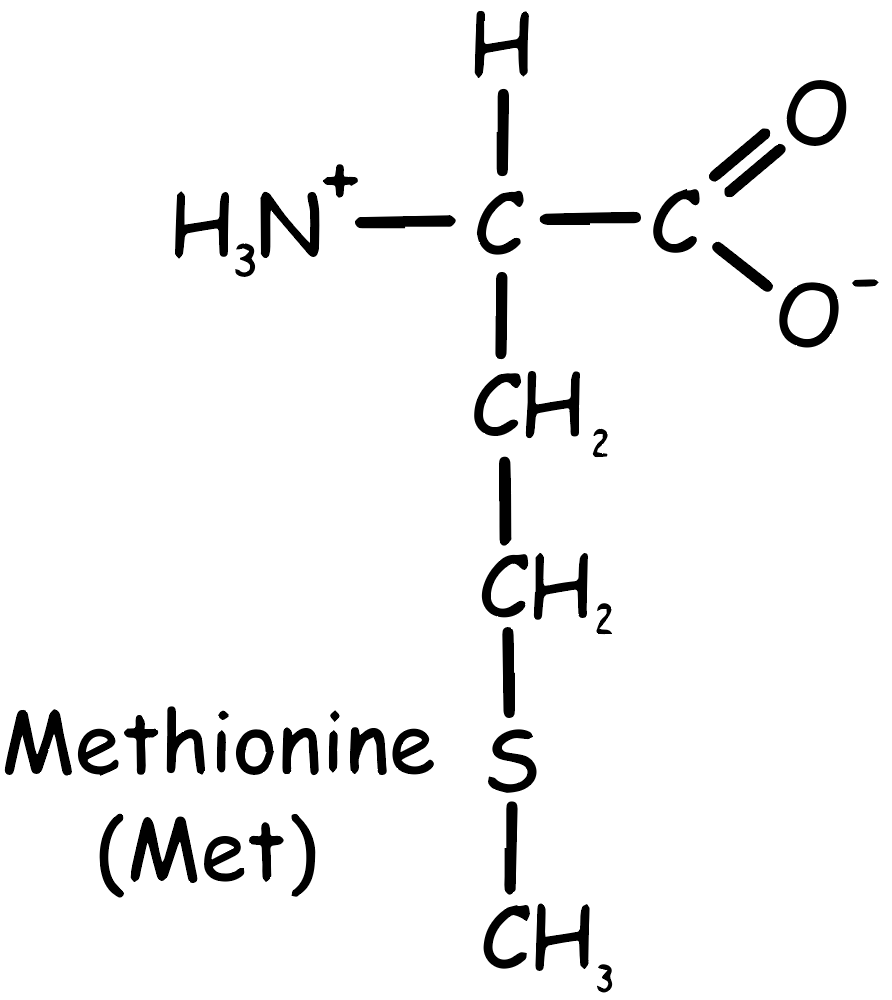
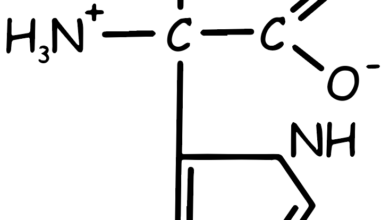Methionine Amino Acid
Methionine is an essential amino acid, meaning it cannot be synthesized by the human body and must be obtained from the diet. It plays a critical role in many cellular processes, including protein synthesis, metabolism, and detoxification.
Chemically, methionine is characterized by its sulfur-containing side chain (see Figure 1), which contributes to its biochemical properties. It is often represented by the single-letter code “M” in protein sequences. Methionine is also important as it is the start codon (AUG) in the genetic code, meaning that protein synthesis usually begins with the incorporation of methionine. This amino acid can be found in various foods, including meats, fish, eggs, and some nuts and seeds.
Methionine is essential but, once inside the body, it can be transformed into Cysteine (another sulfur containing amino acid). Therefore, while Methionine is an essential amino acid, Cysteine is considered non-essential due to this conversion process.
Methionine metabolism
The metabolism of methionine is not only crucial for the synthesis of proteins and other biomolecules but also for maintaining the body’s methyl group balance and sulfur-containing amino acid levels. Any disruptions in this metabolic pathway can have significant implications for health.
As shown in the Figure 2, firstly methionine is activated to form S-adenosylmethionine (SAM), a critical methyl donor in the body. This is done through a reaction with adenosine triphosphate (ATP). SAM donates its methyl group to a variety of substrates, including DNA, proteins, and lipids, in a process essential for many biological functions. After donating its methyl group, SAM is converted into S-adenosylhomocysteine (SAH).
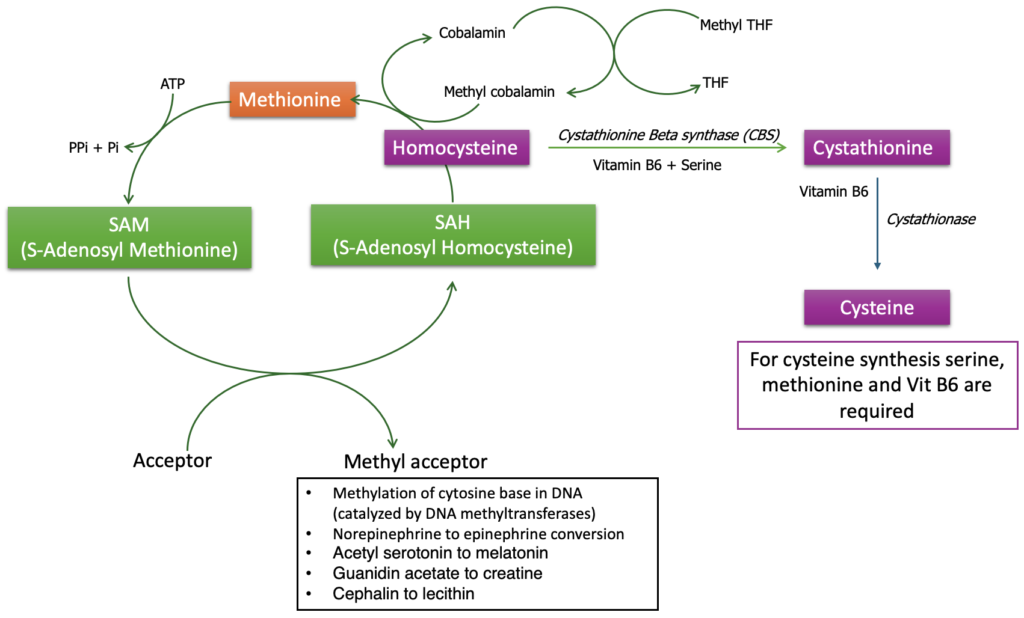
SAH is then hydrolyzed to homocysteine and adenosine. This step is crucial because homocysteine levels are tightly regulated due to its potential toxicity at high concentrations. Homocysteine can be converted into cysteine through the transsulfuration pathway. This conversion requires vitamin B6 (pyridoxine) and involves several enzymes, including cystathionine β-synthase and cystathionine γ-lyase.
Alternatively, homocysteine can be remethylated to methionine. This process requires folate and vitamin B12. The enzyme methionine synthase catalyzes the transfer of a methyl group from 5-methyltetrahydrofolate to homocysteine, regenerating methionine and tetrahydrofolate.
The balance between these pathways is crucial for maintaining proper cellular function and avoiding disorders such as hyperhomocysteinemia, which is associated with cardiovascular diseases.
Homocycteine and cardiovascular disease
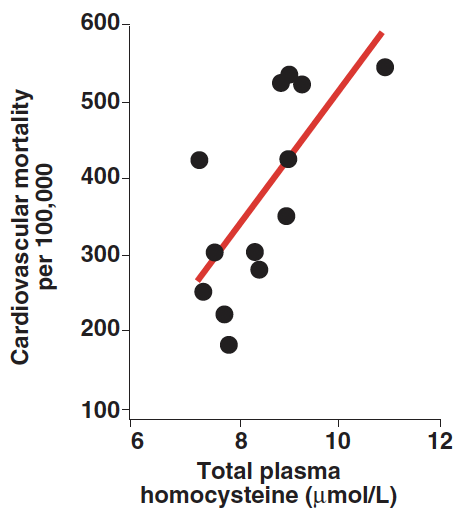
Elevated levels of homocysteine in the blood can lead to oxidative stress, inflammation, and damage to the endothelium, and are considered an independent risk factor for diseases related to blood vessel blockage, as illustrated in Figure 3. Approximately 7% of the population exhibits mild increases in homocysteine levels. Research has demonstrated an inverse relationship between blood homocysteine levels and levels of folate, B12, and B6—vitamins that play a role in converting homocysteine into methionine or cysteine. Taking supplements of these vitamins can reduce homocysteine levels in the bloodstream. However, for patients with existing cardiovascular disease, taking these vitamin supplements doesn’t appear to reduce the incidence of cardiovascular events or mortality. This leads to a question about the role of homocysteine: is it a direct cause of vascular damage, or simply an indicator of such damage?
Note that severe increases in blood homocysteine levels can occur due to rare deficiencies in the enzyme cystathionine β-synthase, as seen in individuals with classic homocystinuria. These individuals often experience early-onset vascular diseases, with about 25% succumbing to thrombotic complications before reaching 30 years of age.
High levels of homocysteine or low levels of folic acid in expectant mothers are linked to a higher likelihood of neural tube defects (like spina bifida, where there is incomplete closure) in the developing fetus. Taking folate supplements around the time of conception has been shown to decrease the risk of these defects.