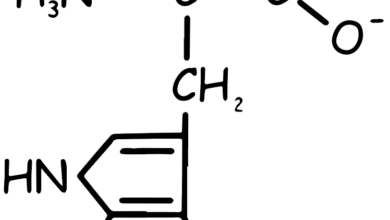
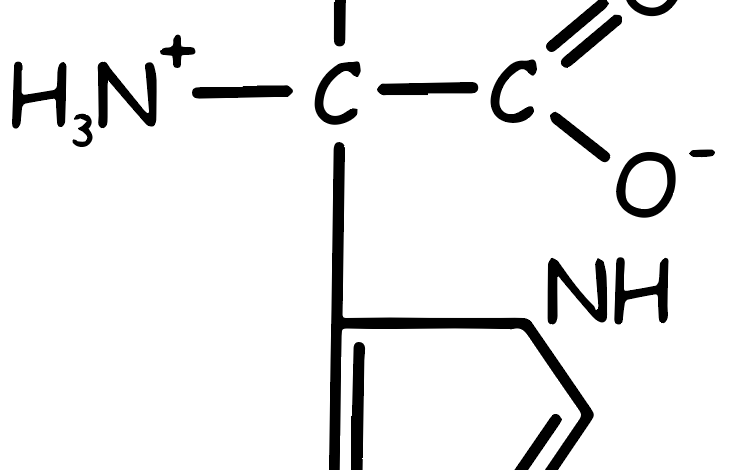
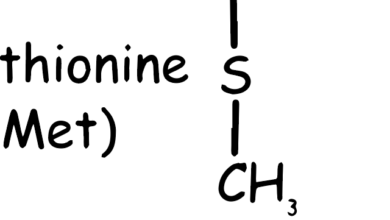Histidine Amino Acid
It has a positive charge, but this is less pronounced compared to other positively charged substances. In terms of polarity, it is indeed polar, yet it exhibits a lower degree of polarity than typical polar compounds. Interestingly, it falls into the category of being essential, yet only partially so, qualifying it more as a semi-essential substance. A notable feature is its maximum buffering capacity. This is primarily due to the presence of histidine, the only amino acid whose side chain can ionize within the physiological pH range, making it unique in this regard. Finally, in its metabolic pathway, it leads to the formation of glutamate via the FIGLU process. This aspect categorizes it as glucogenic, as glutamate is a key metabolite in glucose synthesis. Dietary sources of histidine include meat, fish, dairy products, nuts, seeds, and legumes.
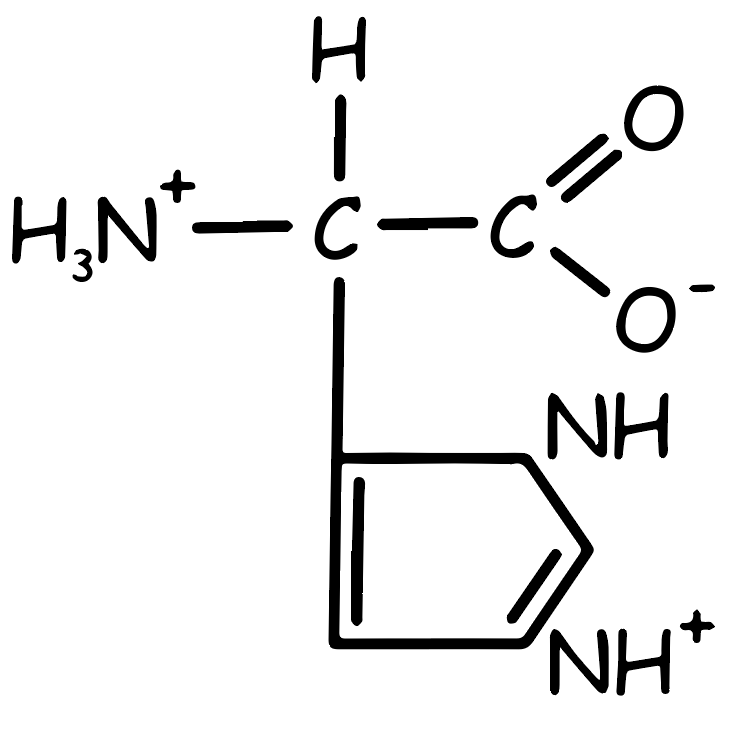
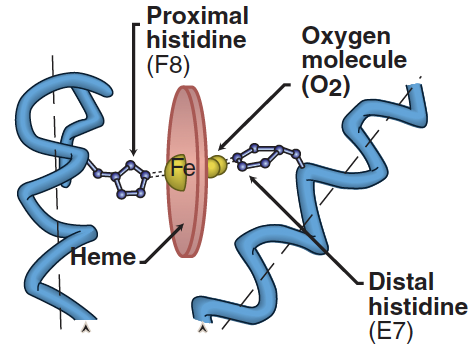
In basic amino acids, the side chains act as proton acceptors. At a physiological pH level, the side chains of amino acids like lysine and arginine are completely ionized and carry a positive charge. However, histidine differs as it is only weakly basic, resulting in its free amino acid form being predominantly uncharged at physiological pH. The unique aspect of histidine is its behavior when it becomes part of a protein. In this context, its side chain can exhibit a positive charge or remain neutral, influenced by the ionic conditions created by the protein’s polypeptide chains. This characteristic of histidine is crucial, as it significantly influences the function of proteins, including hemoglobin and myoglobin (see Figure 2).